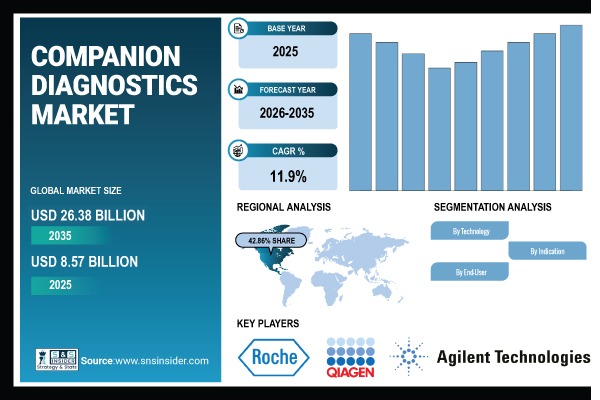
Austin, United States, April 13, 2026 (GLOBE NEWSWIRE) -- As per the SNS Insider report the Companion Diagnostics Market size was valued at USD 8.57 billion in 2025 and is projected to reach USD 26.38 billion by 2035, expanding at a CAGR of 11.9% over 2026–2035. The expanding use of precision medicine, the rising incidence of cancer and genetic disorders, and the growing need for focused therapy methods are all driving the global companion diagnostics market.
Market Size and Forecast:
- Market Size (2025): USD 8.57 Billion
- Market Size (2035): USD 26.38 Billion
- CAGR (2026–2035): 11.9%
- Base Year: 2025
- Forecast Period: 2026–2035
- Historical Data: 2022–2024
Get a Sample Report of Companion Diagnostics Market: https://www.snsinsider.com/sample-request/2915
The U.S. Companion Diagnostics Market was valued at USD 3.41 billion in 2025 and is expected to reach USD 10.52 billion by 2035, growing at a CAGR of 11.95% over 2026–2035. The U.S. continues to lead the global companion diagnostics market, supported by a well-established healthcare infrastructure, strong presence of leading pharmaceutical and biotechnology firms, and early adoption of advanced diagnostic technologies.
Growing Demand for Precision Medicine to Propel Market Growth Globally
The need for companion diagnostics is mostly being driven by the growing trend toward precision medicine, where therapies are customized based on each patient's genetics and biomarker profiles. To assure better treatment success and fewer adverse responses, pharmaceutical and biotechnology companies are increasingly collaborating to create tailored medicines in addition to diagnostic testing. Companion diagnostics play a crucial role in clinical decision-making, and regulatory bodies like the FDA continue to highlight their incorporation into medication approval processes. The usage of companion diagnostic solutions is increasing in oncology, infectious diseases, and other therapeutic areas due to the rising alignment between therapies and diagnostics, which is bolstering the global market trajectory.
Major Companion Diagnostics Companies SWOT Analysis Listed in the Report are
- F. Hoffmann-La Roche Ltd.
- QIAGEN N.V.
- Agilent Technologies, Inc.
- Thermo Fisher Scientific Inc.
- Abbott Laboratories
- Illumina, Inc.
- bioMérieux SA
- Danaher Corporation
- Myriad Genetics, Inc.
- Guardant Health, Inc.
- Foundation Medicine, Inc.
- Sysmex Corporation
- Bio-Rad Laboratories, Inc.
- Hologic, Inc.
- Invitae Corporation
- NeoGenomics Laboratories, Inc.
- Exact Sciences Corporation
- Fujirebio Diagnostics, Inc.
- ArcherDX, Inc.
- Genomic Health, Inc.
Companion Diagnostics Market Segmentation Analysis:
By Technology
The polymerase chain reaction segment held the largest revenue share of approximately 41.82% in 2025, supported by its widespread adoption in clinical diagnostics globally. The next generation gene sequencing segment is expected to expand at the highest CAGR of approximately 12.64% during 2026-2035, driven by increasing demand for comprehensive genomic profiling globally.
By Indication
The cancer segment captured the leading share of nearly 67.35% in 2025, owing to the extensive use of companion diagnostics in oncology for patient stratification, targeted therapy selection, and monitoring treatment response. The infectious diseases are projected to grow at the fastest CAGR of approximately 12.18% over the forecast period, supported by the increasing need for rapid and precise diagnostic solutions globally.
By End-user
Pharmaceutical and biopharmaceutical companies accounted for the largest market share of approximately 46.27% in 2025, driven by the rising integration of companion diagnostics in drug development pipelines and regulatory requirements for targeted therapies. The contract research organizations are expected to record the highest CAGR of approximately 12.52% during 2026-2035, fueled by increasing outsourcing of clinical trials globally.
Do you have any specific queries or need any customization research on Companion Diagnostics Market, make an Enquiry Now: https://www.snsinsider.com/enquiry/2915
Companion Diagnostics Market Key Segments
By Technology
- Polymerase Chain Reaction
- Immunohistochemistry
- In-situ Hybridization
- Next Generation Gene Sequencing
- Others
By Indication
- Cancer
- Neurological Diseases
- Infectious Diseases
- Others
By End-user
- Pharmaceutical & Biopharmaceutical Companies
- Reference Laboratories
- Contract Research Organizations (CROs)
Regional Insights:
North America accounted for the leading revenue share of more than 42.86% in 2025E within the global companion diagnostics market, underpinned by a highly developed healthcare ecosystem, stringent regulatory oversight for precision medicine, and consistent investments in biomarker-driven diagnostic technologies.
With a compound annual growth rate (CAGR) of 12.34%, Asia Pacific is expected to be the companion diagnostics market's fastest-growing region. This is due to the region's continuing expansion of healthcare infrastructure, growing investments in precision oncology, and changing regulatory frameworks in China, India, Japan, and South Korea.
Recent Developments:
- In February 2025, Roche introduced an advanced companion diagnostic assay designed to identify novel biomarker signatures, improving patient stratification and enabling more accurate therapy selection across global oncology treatment centers.
- In June 2024, QIAGEN launched a new NGS-based companion diagnostic panel with expanded biomarker coverage, strengthening its position in precision oncology diagnostics across North America and Europe.
Exclusive Sections of the Report (The USPs):
- PRICING BENCHMARKING & MARKET ACCESS ANALYSIS – helps you evaluate average pricing by technology type, regional price variations, and emerging reimbursement models across key markets.
- REGULATORY & COMPLIANCE FRAMEWORK INSIGHTS – helps you understand approval pathways, clinical validation requirements, government policies, and quality standards in precision diagnostics.
- TECHNOLOGY ADOPTION & INNOVATION TRACKER – helps you uncover trends in molecular diagnostics, biomarker research, patent activity, and advancements in personalized diagnostics.
- CLINICAL PERFORMANCE & OPERATIONAL EFFICIENCY METRICS – helps you assess diagnostic accuracy, test approval trends, turnaround time, and laboratory quality standards.
- INVESTMENT & STRATEGIC COLLABORATION ANALYSIS – helps you analyze funding trends, M&A activity, and partnerships between pharmaceutical and diagnostic companies.
- PRECISION MEDICINE ECOSYSTEM & GROWTH INSIGHTS – helps you evaluate innovation hubs, research expansion, and the growing role of companion diagnostics in targeted therapy development.
Buy an Enterprise-User PDF of Companion Diagnostics Market Outlook Report 2026-2035: https://www.snsinsider.com/checkout/2915
Companion Diagnostics Market Report Scope
| Report Attributes | Details |
| Market Size in 2025 | USD 8.57 Billion |
| Market Size by 2035 | USD 26.38 Billion |
| CAGR | CAGR of 11.9% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
Related Market Reports
Oncology Companion Diagnostic Market
Next-Generation Sequencing Market
About Us:
SNS Insider is one of the leading market research and consulting agencies that dominates the market research industry globally. Our company's aim is to give clients the knowledge they require in order to function in changing circumstances. In order to give you current, accurate market data, consumer insights, and opinions so that you can make decisions with confidence, we employ a variety of techniques, including surveys, video talks, and focus groups around the world.
